 These supernovas forcefully slammed smaller elements together.įor his 1869 periodic table, Mendeleev arranged the elements in order of ascending mass. Heavy atomic nuclei formed as massive, dying stars exploded. Making elements larger than iron required even more cosmic firepower. Those stellar forges also formed the oxygen that we need to breathe. They included carbon, an element essential for all life as we know it. This slowly forged larger and heavier elements. In the center of these stars, intense pressures fused atomic nuclei - the centers of the atoms - creating larger nuclei. Eventually this would create dense, fiery hot furnaces that we know of as stars. Gravity brought these atoms together in ever larger amounts. Just after the Big Bang, the universe was made up of only hydrogen and helium - the two lightest elements. In 2019, the world is celebrating the periodic table in all its forms and how it helps organize and make sense of the building blocks of our universe. Those less traditional periodic tables provide ways not only to highlight some of chemistry’s quirks, he says, but also to bring them into better focus. He’s a chemist at Le Moyne College in Syracuse, N.Y. “Alternate forms are useful because of the different aspects of the science that they illustrate,” notes Carmen Giunta. Scientists and teachers in other fields developed others. Scientists have built many, some with widely varying shapes. It even helps them predict how new materials will behave.ĭmitri Mendeleev/Science History Instituteīut Mendeleev’s well-known chart is far from the only periodic table. It also helps them understand how life works. Understanding those relationships helps chemists create new compounds. How the rows and columns on this table line up points to shared traits between groups of related elements. These patterns link elements with similar chemical behaviors and help to tell chemists how atoms react to form molecules. It holds that shared traits among chemical elements repeat in regular patterns as elements get larger. The rows and columns on the periodic table map the so-called periodic law. /periodic-table-of-the-elements-2017--illustration-769723031-5ac10eb6a9d4f9003769784d.jpg)
Their atoms knit together to form literally everything - us, the air we breathe, the organisms that share our world and every other molecule of gas or bit of mass found throughout our universe. Yet even today, this chart helps scientists make sense of the atoms and molecules that make up our universe.Įlements are the building blocks of all matter. Petersburg, came up with an early version. Dmitri Mendeleev (MEN-duh-LAY-ev), a Russian scientist working in St. This chart - an icon of chemistry - is known as the Periodic Table of the Elements. We created a timeline of the history of the periodic table.Hanging on the wall, the squares look like a jagged wall of alphabet blocks. The Periodic Table has constantly been improved and developed over the past 200 years, but in 1869 Dimitri Mendeleev finished the first version of the periodic table as we know it today, by arranging the elements by atomic mass and leaving spaces open for the elements that were not yet discovered. Who invented the Periodic Table of Elements? The periodic table also gives us an idea of what the characteristics of an element might be and help us predict how an element might react based on in which group it is located. 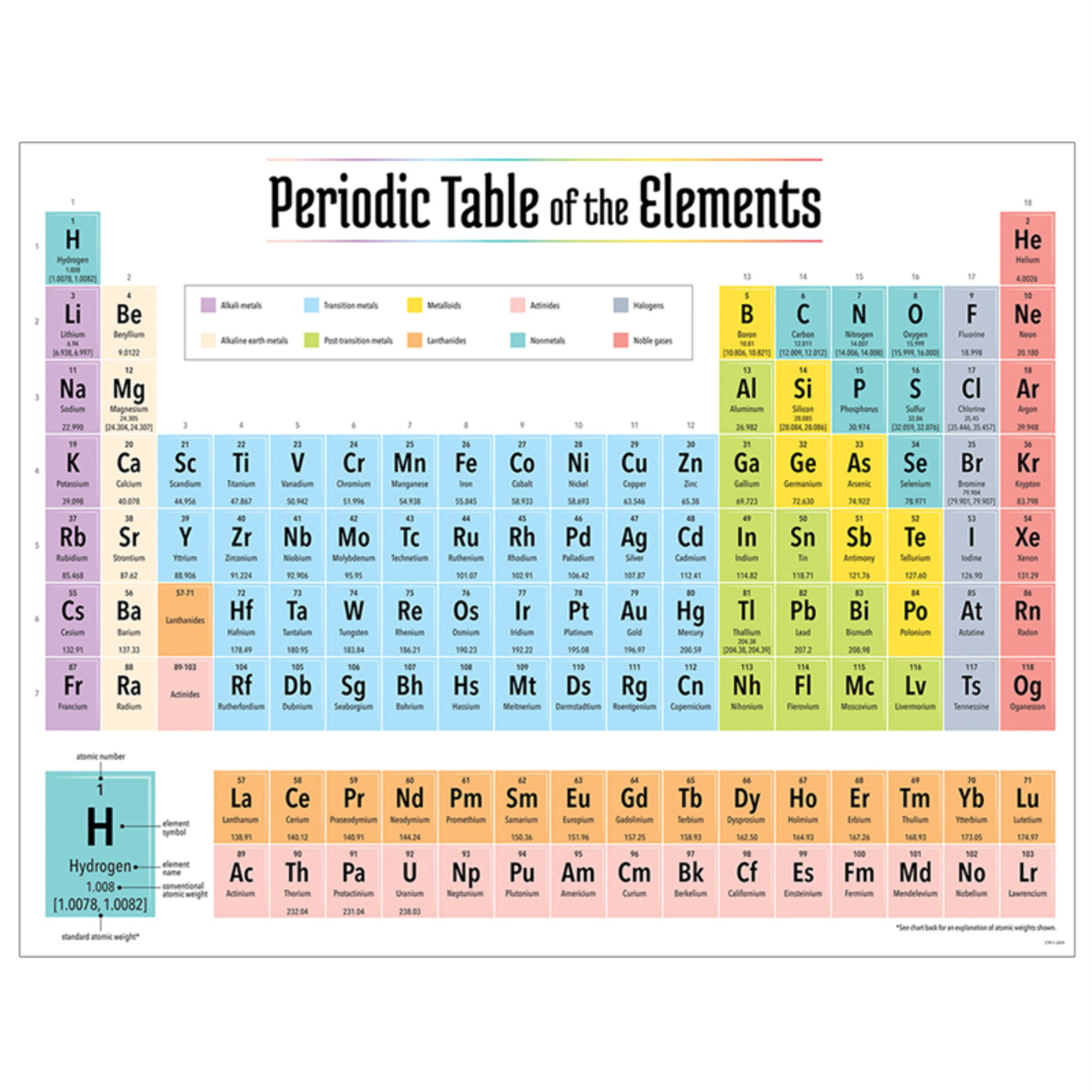 
The Periodic Table of Elements can be used as an assisting tool in chemical calculations, when a specification of an element is needed it is easily found in the Periodic Table. How is the Periodic Table of Elements used? The table lists all the elements that are currently known (118), in descending order of the number of protons that are present, in a single atom of the element. The Periodic table of elements is a tool, developed by scientists over hundreds of years. List of all the elements and their properties: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
